Phase I: Concept development of diagnostics and medical products
Your PLUS with Gerresheimer
Development of all types of ideas and product concepts
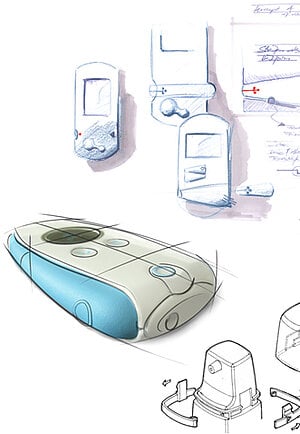
Gerresheimer would like to already plan the initial product ideas together with you. In the first step, we start with a thorough analysis of the market, the needs of the users, and the competition. We concentrate on all information of relevance to the product and carry out user surveys. We understand the market and user requirements for your product idea, identify, prioritize, and implement the user requirements in corresponding design guidelines. Thanks to incorporation into the early phases of concept development, the Gerresheimer team can then develop concepts that not only take these market and user requirements into account, but are also oriented to subsequent production. By way of prototypes from the 3D printer, which can be used early on in the development process, both individual product functions and overall concepts can be tested directly and the feedback is incorporated into the development process.
Industrial design
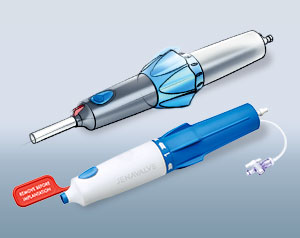
Our product designers in Münster have more than 15 years of experience in the design of diagnostics and medical products. They work with a clear vision: form follows function. The design must improve the functionality of the products, consider the needs of the users and patients and design the production processes to be as efficient as possible. At the same time, however, the product design should reflect the distinctive image of our customers and ensure easy recognizability.
Patent management

In the case of product development for diagnostics and medical products, strategic patent management is of essential importance. This involves not only the securing of all created ideas in accordance with patent law. Through targeted patent research, Gerresheimer is able to recognize trends and systematically exploit all opportunities to realize the innovative competence of our customers and our company into a permanent market lead.
Development of operating concepts as part of usability engineering
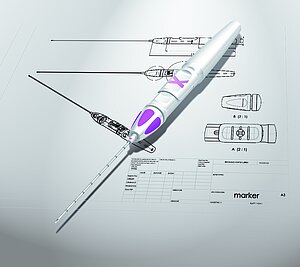
We combine market needs with the functional and technical requirements of the diagnostics and medical product. This encompasses not only an attractive design, but also and especially ergonomics suited to the function. Especially in the case of diagnostics and medical products, the intuitive, user-friendly, and easy handling of the product is a high priority. The focus of our development is always on future users: patients, medical personnel, or pharmacists, with their daily tasks, needs, and sensitivities. Through direct contact with them, we acquire especially valuable knowledge from routine practice and transfer this to our concepts. We develop the most varied operating concepts with the goal of considering and minimizing risks and operating errors in subsequent usage.
Evaluation of the design concepts as part of usability studies

We evaluate several alternative design concepts in the context of user studies. This gives us and our customers a sense of security of having chosen the best design concept and of having minimized risks and operating errors in subsequent use. This design variant is then further optimized in the following phases and realized up to the stage of clinical samples. Our customers receive this process in the form of documentation in keeping with standards, which enormously accelerates the approval process for medical devices.
Concept evaluation and feasibility studies
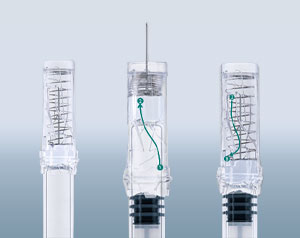
Various approaches to solutions are then available in the form of concepts at the end of Phase I. Each concept is evaluated in close cooperation with the customer. Depending upon the project goal, the requirements of the product and the feasibility with regard to production in the desired number of units is checked in the process, and the practicability of the required processes and production technologies is evaluated at an early date. The evaluation can also take place using mock-ups, which are already very similar to the subsequent product in terms of look, feel, and function. New ideas can still be easily realized at this point in time.

